Britain today secured an additional 4.25million courses of at-home Covid pills to shield against the Omicron variant this winter.
Included in the order are 2.5million more courses of Pfizer’s Paxlovid, which has been shown to slash the risk of vulnerable adults being hospitalised by up to 90 per cent.
The Government has also ordered another 1.75million courses of Lagevrio from Merck, shown to cut the death rate by about 30 per cent.
While these studies looked at older variants, manufacturers say the pills should still work well against Omicron because they do not target parts of the virus prone to mutations.
They are both given within five days of a positive test and will be targeted at the most vulnerable.
Health Secretary Sajid Javid hailed the move as a ‘mammoth’ deal. The orders will arrive in early 2022.
The order is on top of 480,000 doses of Lagevrio — the brand name for molnupiravir — and 250,000 of Paxlovid purchased in October.
Lagevrio is already being rolled out on the NHS to immunocompromised patients, cancer sufferers and other people at high risk of severe Covid.
Paxlovid could be given the green light from Britain’s medical regulators within weeks and will be deployed in the same way.

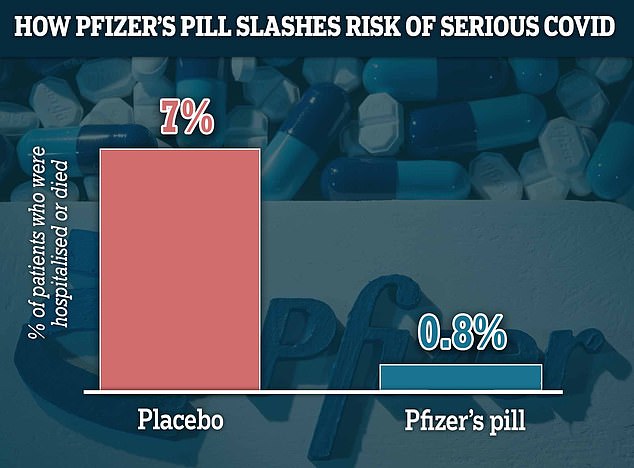
Pfizer’s twice-a-day antiviral was shown in clinical trials to slash the risk of hospitalisation or deaths by 87 per cent in vulnerable and elderly patients. It found that 0.8 per cent of those given Pfizer’s drug within three days of symptom onset were hospitalised and none had died by 28 days after treatment. For comparison, the hospitalisation rate stood at 7 per cent for the patients given a placebo. There were also seven deaths in the placebo group

Lagevrio was shown to cut hospitalisations and deaths by up to 50 per cent in early trials but Merck later revised this to 30 per cent. It works by disrupting the Covid virus’s ability to reproduce in the human body
Health chiefs have not revealed the cost of the deal, but previously the Lagevrio pills were secured at a cost thought to be in the region of £250million.
It is not clear how much was paid for the Paxlovid pills, but Doctors without Borders says it costs about £525 ($700) for one course of the treatment.
Taken together, they suggest the Government has paid more than £1billion for the extra courses of each treatment.
Last week, Lagevrio was made available on the NHS to the most high risk patients who have tested positive for Covid.
It is also being rolled out through the national Panoramic study, which was set up by Oxford University to test antivirals.
Paxlovid — which is the brand name for drugs PF-07321332 and antiviral ritonavir — will be added to the trial once it is approved by regulators.
The trials aim to recruit 10,000 elderly people and those who are more vulnerable to Covid, with ministers calling on everyone eligible to come forward.
Sources said the treatments could be made available on the NHS early next year once data is available from the study.
Paxlovid should be taken at home within five days of testing positive.
Medics say people receiving this treatment should take two pills twice a day for five days. One pill contains the drug PF-07321332, and the other contains ritonavir.
The treatment is a combination of an experimental drug with an older antiviral called ritonavir, which is already used to treat HIV/AIDS.
Pfizer’s drug, part of a class known as protease inhibitors, is designed to block an enzyme Covid needs in order to multiply.
It is given with ritonavir to make sure it stays active in the body for a longer period of time.
Lagevrio is also taken at home, and should be administered within five days of a positive test. Patients are told to take the pill twice a day for five days.
Developed by German pharmaceutical giant Merck in partnership with Ridgeback Biotherapeutics, it works differently to Paxlovid and is designed to introduce errors into the genetic code of the virus.
Both pharmaceutical giants say their pills should be equally effective at tackling new variants.
Vaccines, which are being made slightly weaker by new strains of the virus, work by targeting the spike protein and were designed to tackle the original virus.
Because a lot of evolution happens on the spike protein, the immune systems of vaccinated people sometimes find it more difficult to recognise new variants.
But the pills should provide an extra layer of protection on top of vaccine-induced immunity to protect against the virus.
Mr Javid said: ‘Our Covid booster programme continues at unparalleled pace and it’s vital we further bolster our national response to the virus by ensuring access to the world’s best treatments too.
‘This is a mammoth deal for the UK Government and for patients across the country that are set to benefit from these antivirals over the coming months.
‘If you’re aged 50 and over or have an underlying health condition and test positive for Covid, sign up to the study as soon as you can and take advantage of this remarkable treatment.’
Antivirals Taskforce chairman Eddie Gray said: ‘Both antivirals in our current portfolio could be vital tools against Omicron – with current evidence showing they will be effective against the variant.
‘The Antivirals Taskforce will continue its work to ensure vulnerable people who test positive for Covid-19 can rapidly access these medicines.’
Last month the UK became the first country in the world to approve the use of Lagevrio in patients, in a move heralded as a ‘game-changer’ by Mr Javid.
Merck initially said its pill slashed the risk of hospitalisation and death by half in vulnerable patients.
But new data from the second arm of the study only recently published showed it did not improve outcomes for those that received it compared to the placebo group.
French medical regulator’s refused to approve the pill last week, saying it was less effective than initially hoped.
The US paid $1.2billion (£900million) for 1.7million courses of Lagevrio in June.
Its drug regulator, the FDA, is expected to approve the treatment within days.