The Food and Drug Administration (FDA) has expanded the availability of the COVID-19 vaccine booster shots in the wake of new data showing the Omicron variant can bypass protection provided by the first two doses.
The agency announced Thursday that it has expanded the eligibility of the Pfizer-BioNTech booster for people 16 and 17 years old.
It is the youngest age group to become eligible for the booster shot in America.
All Americans 16 or 17 will be able to receive the additional shot six months after completion of their initial vaccine series.
On Wednesday, Pfizer unveiled data showing that people who had received only the first two-shots were vulnerable to infection from the Omicron variant, but that the third shot could return protection back to original levels.
The company did not say whether the first two doses alone was enough to prevent sever illness caused by Covid.

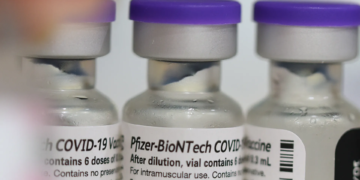
The Pfizer COVID-19 booster has receive authorization from the FDA to be used in Americans aged 16 and 17 (file photo)

The Pfizer vaccine is the only jab available to minors in the U.S. Recent data revealed by the company finds that the initial two-dose vaccine series is less effective against the Omicron COVID-19 variant, but receiving a booster shot could increase antibody levels 25-fold. Pictured: A young woman in Philadelphia, Pennsylvania, receives a shot of a COVID-19 vaccine on August 4

An African research team found that the Pfizer vaccine provided 40 times less Covid antibodies to fight against the Omicron strain that it does against other variants, hinting that it may be less effective at preventing infection
‘Vaccination and getting a booster when eligible, along with other preventive measures like masking and avoiding large crowds and poorly ventilated spaces, remain our most effective methods for fighting COVID-19,’ said Dr Janet Woodcock, acting commissioner of the agency in a statement.
‘As people gather indoors with family and friends for the holidays, we can’t let up on all the preventive public health measures that we have been taking during the pandemic.
‘With both the delta and omicron variants continuing to spread, vaccination remains the best protection against COVID-19.’
On Wednesday, Pfizer announced that preliminary data as to how its vaccine responds to the Omicron variant.
The company announced that it had data showing its initial two-dose vaccine regimen was not as effective against the new strain of the virus as it was against others.


It came a day after the African Health Research Institute revealed pre-print data showing the vaccine had 40 times less antibodies effective against the Omicron variant than it had against others.
Pfizer said that receiving a booster, though, would increase the available antibodies for fighting Omicron 25-fold.
‘Although two doses of the vaccine may still offer protection against severe disease caused by the Omicron strain, it’s clear from these preliminary data that protection is improved with a third dose of our vaccine,’ Albert Bourla, CEO of Pfizer, said in a statement.
‘Ensuring as many people as possible are fully vaccinated with the first two dose series and a booster remains the best course of action to prevent the spread of COVID-19.’
The variant has been sequenced 71 times in 21 U.S. states as of Thursday afternoon, though the actual number of cases circulating is likely much higher.
It has also been sequenced more than 2,300 times worldwide in around 60 countries.
The FDA reports that myocarditis and pericarditis, both types of health inflammation, are the most common negative side effects reported during clinical trials.
While younger people are at a higher risk of developing the condition after vaccinated, regulators say the benefits of the shots outweigh the risks.
The Pfizer jab is the only one available to minors in America for the first two doses, and is also the most popular vaccine in America.


According to official data from the Centers for Disease Control and Prevention, 61 percent of people between the ages of 12 to 17 have received at least one dose of the Pfizer vaccine, and 52 percent are fully vaccinated.
‘The Pfizer-BioNTech COVID-19 Vaccine has been available to individuals 16 years of age and older for nearly a year, and its benefits have been shown to clearly outweigh potential risks,’ said Dr Peter Marks, director of the FDA’s Center for Biologics Evaluation and Research in a statement.
‘Since we first authorized the vaccine, new evidence indicates that vaccine effectiveness against COVID-19 is waning after the second dose of the vaccine for all adults and for those in the 16- and 17-year-old age group.
‘A single booster dose of the vaccine for those vaccinated at least six months prior will help provide continued protection against COVID-19 in this and older age groups.’


The jab has been administered more than 275 million times since it first became available almost exactly one year ago.
It has also been used to fully vaccinate more than 110 million people, and 26 million Pfizer booster shots have been adminsitered.
Moderna and Johnson & Johnson booster shots are still only available to people 18 and older, though minors who have received those shots are allowed to mix-and-match and receive the Pfizer booster.
Neither of the other two vaccine manufacturers have released data on their vaccine’s ability to prevent infection from the Omicron variant.
A study by National Institute for Health Research (NIHR) in England found that booster doses increase the T-cell response against the virus, which protect a person from severe infection even after infection.









:quality(70)/cloudfront-eu-central-1.images.arcpublishing.com/irishtimes/CGYPT6WE7VAO3KJ773SHUVMVBY.jpg?resize=1200,630&ssl=1)