Covid cases in America have reached one of the lowest points of the pandemic so far, but with fears of the BA.2 ‘stealth’ variant rising in some parts of the country, the Food and Drug Administration (FDA) is expected to approve a fourth dose of the vaccine for older Americans.
CNN reports that the FDA plans to authorize a second booster dose of both the Pfizer-BioNTech and Moderna vaccines, each would be the fourth dose of the company’s vaccine regimen. The Centers for Disease Control and Prevention (CDC) is then expected to issue a recommendation to all Americans 50 and older to receive the shot.
Once the jab receives approval, any American would be eligible to receive it if they wanted but the government will only recommend it to older age groups for now. A similar process played out with the first booster shot, where it was first only recommended to older age groups before the Omicron variant arrived in late 2021.
Whether or not Americans will be up for another booster shot is still to be seen, though. The nation’s booster shot rollout has cratered in recent weeks, with cases plummeting and many looking past COVID-19, hoping the Omicron wave will be the final one of the pandemic.

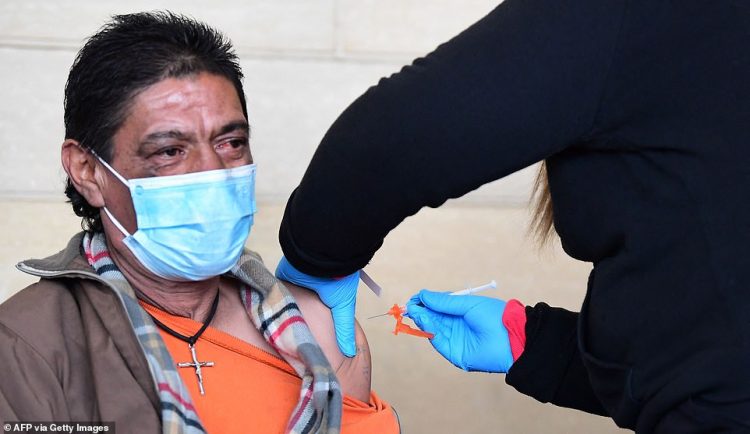
The FDA and CDC plan to soon recommend and then approve COVID-19 vaccine booster shots to Americans aged 50 and older, CNN reports. Pictured: A man in Los Angeles, California, receives a shot of a COVID-19 vaccine



Covid cases have stabilized after a winter that saw dramatic case rises followed by case falls. The nation is currently averaging 31,604 cases per day as of Monday morning, about the same total as the previous week.
Some specific areas of America are seeing worrying increases, though. In Kentucky, cases are up 110 percent over the past two weeks, the first state to record a doubling over that period since early-February. New York, along with eight other states, have seen cases jump over the past two weeks as well.
These cases are not translating into deaths during this surge at the rate that they were previously. America’s high vaccination rate, mixed with the relatively mild nature of the dominant Omicron variant, has managed to protect Americans from the worst outcome.
America is averaging 850 daily Covid deaths as of Monday morning, a 20 percent drop over the past week, and a figure that may continue to trend downwards.
With deaths falling and interest and fear of the pandemic waning across the country, some experts fear it could be challenging to convince Americans to get an additional shot.
Not all experts believe the shot is necessary either.
Dr Anna Durbin is an international public health expert at Johns Hopkins University in Baltimore, Maryland, and has been a critic of Pfizer, Moderna and the White House’s insistence to roll out COVID-19 booster shots before they are needed. Last week, she told ABC that she does not believe many Americans will benefit from additional shots.
‘There are very few, if any, people who, in my opinion require a fourth dose,’ she said.
In August, when the White House was laying out plans to roll out the first batch COVID-19 booster shots, Durbin was also a critic, telling DailyMail.com that there was little science backing up the decision.


A very small number of Americans are already eligible for fourth COVID-19 shots. The Centers for Disease Control and Prevention (CDC) advises people who are immunocompromised to receive the additional shot now, despite the lack of authorization from the Food and Drug Administration (FDA).
Only around one in every 30 Americans is immunocompromised and is eligible for that fourth shot right now, though.
‘In general, it’s too early to recommend a fourth dose, except for those who are immune compromised,’ Dr Paul Goepfert, professor of medicine at the University of Alabama at Birmingham, told ABC.
The next shot is being rolled out earlier than expected out of fear of the BA.s ‘stealth’ variant, a lineage of Omicron.

The ‘stealth’ variant, which earned the moniker from its ability to avoid detection through some sequencing methods, is believed to be the most infectious version of Covid yet – but is just as mild as the BA.1 version of Omciron that took over the world last last year.
According to the most recent data revealed by the Centers for Disease Control and Prevention (CDC) last week, BA.2 makes up 35 percent of active Covid cases in the U.S., with BA.1 still being dominant.
BA.2’s share of Covid infections in America is growing, though, with the variant only accounting for 23 percent of cases in the week previous.
Moderna CEO Stephane Bancel said last week that he expects the U.S. to suffer a BA.2-fueled surge sometime soon, though, and that his company’s vaccine will be needed to control it.
‘Already several countries around the world have some of the 4th dose testing in people at high risk,’ Bancel told CNBC’s Squawk Box. ‘There’s a big wave of BA.2 variant in Europe right now, as many public health experts have said this should start in the U.S. very soon.’


A growing list of experts are saying the exact opposite, though.
‘I would not be surprised at all, if we do see somewhat of an uptick,’ Dr Anthony Fauci, the nation’s top infectious disease expert and someone who has frequently been among the more cautious voices during the pandemic, said at a Washington Post event this week.
‘I don’t really see, unless something changes dramatically, that there would be a major surge.’
While the stealth variant has failed to make a major impact yet on case numbers, data from overseas – referenced by Bancel – is cause for some concern.
Some countries that had experienced declining cases for months, like the UK, France and Denmark, suddenly saw infection rates start to surge last week. Cases seem to have stabilized in these nations and the growth has stopped for now, though.
Internationally, the World Health Organization (WHO) reports that there were over 12 million Covid cases globally last week, a seven percent jump from the previous week.
Deaths dropped, though, down 23 percent to under 33,000 – another sign of the virus’s falling mortality.
The increase in cases was entirely clustered in the Western Pacific region, where daily infections jumped 23 percent last week. In Europe, infections stabilized after slightly rising two percent last week.
Despite falling case numbers and the failure of BA.2 to cause a major surge in the U.S. like it has in many other countries, the fourth shot of a COVID-19 vaccine was likely always on the way.
Fauci, Bancel and Pfizer CEO Albert Bourla have been among those saying an additional dose was on the way for months, with Bourla even saying annual jabs will be needed for the next decade to control the pandemic.


While the shots have been deemed safe and effective by health officials around the world, and have likely saved millions of lives over the past year, Pfizer and Moderna’s goals in the vaccine rollout are not exactly humanitarian.
The companies have each made billions off of the sales of vaccines to the U.S. and other nations around the world.
Pfizer, its partner BioNTech, and Moderna estimate a combined $50 billion in COVID-19 vaccine sales this year, and those figures will soar even higher if fourth doses are approved.
Both companies also are hoping to roll out jabs to young children in the near future. Currently, the Moderna shot is only available to adults in the U.S., with Pfizer’s available to those five and older.
On Wednesday, Moderna announced that it had successfully completed Phase 2 and 3 clinical trials for its COVID-19 shot in children aged six months old to 17.
The shots, which are a quarter of the size of those given to adults, proved to be around 40 percent effective at preventing infection from the Omicron variant – similar to protection levels it provides adults.
Pfizer has run into some issues in getting its vaccine out to the youngest age groups. The company had to shift its Covid vaccine regimen for the youngest children up to three doses from two, as the smaller, three microgram doses, were almost entirely ineffective in children three and four years old.
The New York City-based firm also submitted data to regulators for its Covid jab in children under the age of five, though the approval process was paused earlier this year.