New research conducted by researchers in the lab of Penn’s E. James Petersson in collaboration with Oregon State University and the University of Washington describes how proteins in living cells can be engineered to include synthetic fluorescent amino acids that are bright, long-lasting, and have properties that sense their environment. This work can help biologists study proteins more easily, with implications for understanding the mechanisms of complex neurological diseases. The results were published in Chemical Science with two associated studies published in eLife and Scientific Reports.

New research describes how proteins in living cells can be engineered to include synthetic fluorescent amino acids that are bright, long-lasting, and have properties that sense their environment. Image credit: E. James Petersson
The Petersson lab has long been interested in developing new ways to label proteins with fluorescent tags, especially for proteins like tau- and alpha-synuclein, which are implicated in Alzheimer’s and Parkinson’s diseases. Currently, specific proteins are tracked in cells by adding fluorescent tags like green fluorescent protein (GFP) and using microscopy to find their location in cells. However, proteins like GFP are very large, making it challenging to study smaller proteins, including alpha-synuclein, which is around half the size of GFP. Other platforms with smaller tags do exist, but because they are toxic they cannot be used to study living cells for long periods of time.
An alternative approach being pursued by the Petersson lab is to chemically synthesize fluorescent versions of amino acids, the small, individual building blocks of proteins. Then, researchers can use engineered enzymes that allow the fluorescent amino acid to be incorporated into proteins when they are being made in a cell. “By encoding our fluorophores as amino acids, our system can give you a labeled protein that behaves exactly like the native protein,” says Petersson.
Previously, the Petersson lab had been able to get bacteria to add an amino acid not found in nature called acridonylalanine (Acd) into its proteins, but they hadn’t yet been able to get this system to work in mammalian cells, which are more relevant to studying human disease. Now, with the help of collaborators at Oregon State University and the University of Washington, first author and Petersson lab Ph.D. student Chloe Jones was able to bring together a breadth of techniques spanning synthetic organic chemistry, molecular biology, cell biology, and microscopy in order to incorporate Acd into proteins in mammalian cells.
First, the researchers conducted directed evolution experiments in E. coli in the lab of Ryan Mehl, a leader in genetic code expansion at Oregon State. Using engineered aminoacyl tRNA synthetase enzymes, the researchers were able to “hijack” the protein synthesis process by reassigning a portion of the E. coli genetic code to be designated as a place where Acd should be inserted. Then, the researchers determined which specific mutant synthetase enzymes were the best for incorporating Acd into the protein structure. They also ran computational models to understand why the specific mutations made this possible.
After refining their methods for incorporating Acd into proteins in E. coli, the researchers relied on the expertise of the University of Washington’s William Zagotta and Sharona Gordon, who had experience adding unnatural amino acids into mammalian cells, most notably on neuronal ion channel proteins.

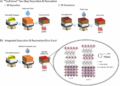
Depictions of the protein structures of green fluorescent protein (far left) compared to alpha-synuclein (top) along with the engineered amino acid acridonylalanine (Acd) and the natural amino acid tryptophan (Trp). The inset image (bottom right) shows how this synthetic fluorescent amino acid can be used to image alpha-synuclein proteins in living cells. Image credit: E. James Petersson