Mind-reading brain implant allows paralysed man, 34, who can’t speak or move to communicate with family for the first time in years – and one of his first requests is for a BEER
- A completely paralysed German man had two electrodes inserted into his brain
- The man had been unable to communicate with his family since February 2019
- After months of training the man was able to convey yes or no through a speaker
- He can now ask for a beer and ask his son if he would like to watch a Disney film
A paralysed man unable to even move his eyes can now communicate thanks to a brain implant — and one of his first requests was for doctors to fetch him a beer.
The unidentified German man, 34, could also tell his four-year-old son he loved him and ask if he would watch a Disney film with him.
He has been in a completely ‘locked-in’ state since February 2019 due to the progression of his amyotrophic lateral sclerosis (ALS).
The disorder, also known as motor neurone disease, affects the brain and the nerves governing movement. Over time, it can cause paralysis.
As it progresses, sufferers are gradually robbed of verbal communication, relying on eye movements to convey ideas to their loved ones.
But even this can be eventually stripped away from them, as nerves governing these movements also degenerate.
However, scientists say the new communication method could offer hope to thousands living with the condition allowing them to to spell out words to loved ones.

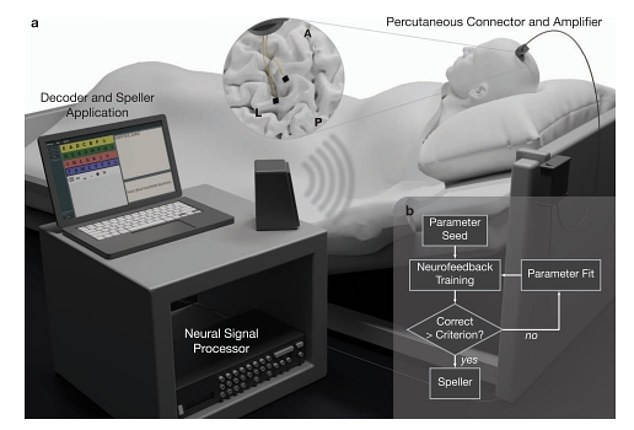
This graphic demonstrates how the system works. Electrodes implanted into the man’s brain pick up on signals attempting movement which are translated into yes or no responses. These are communicated back to the man through a speaker which emits one tone for yes and a different tone for no. Through this yea and no system the man can spell out words and communicate requests and feelings to people
Publishing their findings in the journal Nature Communications, medics surgically inserted an implant consisting of two small electrodes into the man’s motor cortex, the part of the brain responsible for movement.
Over the course of two months, the researchers monitored activity in the region while asking him to simply think about moving various parts of his body.
He was then trained to use thinking about specific movements to answer ‘yes’ or ‘no’ to questions.
The system allowed him to communicate words and ideas one letter at a time — at a rate of about once character a minute.
After three-and-a-half months of living with the brain implant, the man was able to communicate short phrases.
His first achievement was spelling his own name, followed by his relative’s.
He then thanked the team of medics and technicians who gave him back a means of communication.
The patient then gradually began to use the system to improve his care and make requests.
On one occasion, the man asked to be given a beer through his feeding tube.
He was also able to tell his child ‘I love my cool son’ and directly ask if he would like to watch a Disney film.
One of his most repeated requests, the authors noted, was to ask for his family play an album by American rock band Tool ‘loud’.
Professor Niels Birbaumer, a neuroscientist at the University of Tübingen, revealed the paralysed man thanked him and his team directly.
ALS patients who still have control of their eye movements are able to rely on a eye or facial muscle based systems of communication which can be used to spell out words and ideas using dedicated tracking software, with relatively fast speed
Professor Birbaumer claimed it would be valuable to test the implant on more people paralysed with advanced ALS.
About 30,000 Americans are estimated to have ALS, with 5,000 new cases diagnosed each year.
In the UK, an estimated 5,000 people are estimated to be living with motor neuron disease at any one time.
ALS sprang to viral global fame during the Ice Bucket Challenges, where participants doused themselves in frigid water to fundraise for the disease.
Physicist Sir Steven Hawking famously had ALS and managed to beat the limited lifespan that many people have, surviving until the age of 76.
The study does not state when the man became paralysed but it does say he became unable to move his eye muscles in February 2019 which he previously used to communicate.
Advertisement








:quality(70)/cloudfront-eu-central-1.images.arcpublishing.com/irishtimes/CGYPT6WE7VAO3KJ773SHUVMVBY.jpg?resize=1200,630&ssl=1)