Solar energy is getting cheaper and cheaper. However, some challenges are still standing. For example, how do you store that energy preparing it for when it is actually going to be useful? Because the sun doesn’t usually shine when we need the electricity. We need cheaper batteries and now researchers at Cornell Universityfound a way to make cheap batteries that would last for a long time.

Aluminium particles deposited on carbon fibre. These batteries could last a long time. Image credit: Cornell University
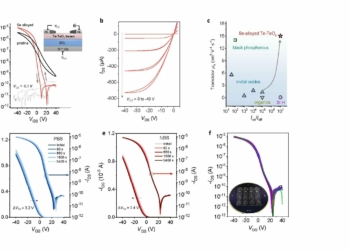
The problem with our current battery technology is that they use expensive materials. Meanwhile aluminium is relatively cheap, because it is found commonly in Earth’s crust. Furthermore, it is already used very widely, which means that the aluminium industry is well-developed and can supply a lot of metal very quickly and cheaply. However, until now aluminium-anode batteries were unsuccessful, because this metal chemically reacts to fibreglass substrate separating the two electrodes. This means that anode would get eaten away quickly and the battery could short of fail in another way.
Now scientists solved this problem with some creative thinking. They used a carbon fibre substrate to separate anode and cathode. Aluminium anode still erodes, but not as quickly. When the battery is charged, aluminium reacts to the carbon structure – these two materials begin exchanging electron pairs. In other words, aluminum is deposited into the carbon structure via covalent bonding. However, another trick prevents this battery from failing too quickly – instead of a conventional two-dimensional architecture for electrodes it used a nonplanar structure.
This means that aluminium anode is constructed from thicker layered metal, which can be finely controlled and the electrode has much faster kinetics. Lynden Archer, senior author of the study, said: “Although superficially different from our earlier innovations for stabilizing zinc and lithium metal electrodes in batteries, the principle is the same – design substrates that provide a large thermodynamic driving force that promotes nucleation; and runaway, unsafe growth of the metal electrode is prevented by forces such as surface tension that can be massive at small scales”.
Because aluminium is cheap, these batteries would be affordable. But that doesn’t mean that these systems would need a lot of them or a tonne of maintenance. Scientists tested aluminium-anode batteries and found that they offer up to 10,000 error-free cycles.
Of course, they do not offer as high of a capacity as conventional lithium-ion batteries, but that’s not the point. Cheaper aluminium-anode batteries could help us collect solar energy and use it later. This kind of technology is only getting better and at the same time it is driving the cost of solar power down.
Source: Cornell University