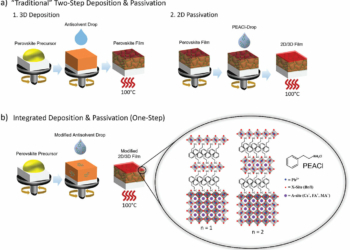
We know sodium is a chemical element. It belongs to alkali metal group and is the sixth the most abundant element in the Earth’s crust. Despite its abundance, it is not found in the nature in its pure form due to a very high reactivity with other elements.

Spinning sphere of molten sodium is actually used in a real scientific experiment. Could you guess what is its purpose? (Still image from YouTube video)
We also know that sodium forms a part of different useful chemicals. It is even possible to manufacture table salt using reaction between sodium and chlorine. And there is also this huge spinning sphere of molten sodium. Why in the world we would need that??
The following video explains the details of this scientific experiment, which actually has a very real practical purpose: